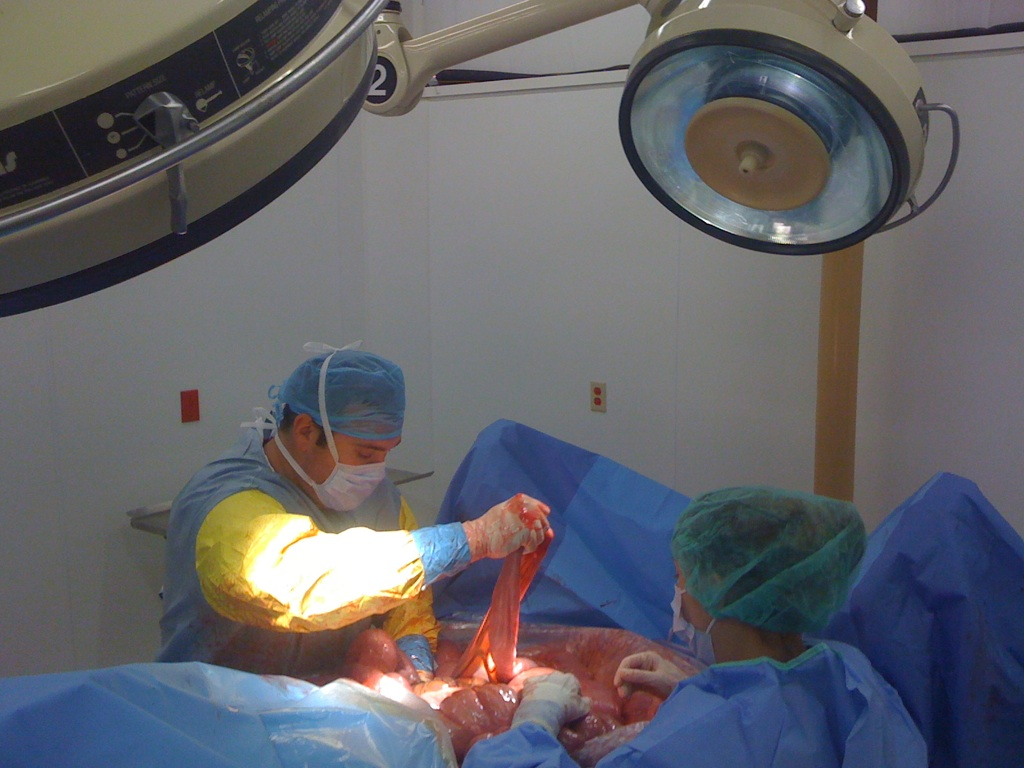
CALEC surgery, or cultivated autologous limbal epithelial cells, represents a groundbreaking advancement in the field of eye damage treatment, particularly for patients suffering from severe corneal injuries. Conducted at the prestigious Mass Eye and Ear, this innovative procedure utilizes stem cell therapy to restore the corneal surface by transplanting healthy limbal epithelial cells from an unaffected eye. With more than a 90 percent success rate observed during clinical trials, CALEC surgery has become a beacon of hope for those facing vision impairment due to corneal damage. This pioneering approach not only highlights the remarkable potential of stem cell technology but also paves the way for future advancements in corneal restoration. As researchers continue to refine this technique, the possibility of expanding its applications could transform the landscape of ophthalmic care worldwide.
Cultivated autologous limbal epithelial cell transplantation, commonly referred to as CALEC surgery, signifies a monumental breakthrough in ocular therapeutics, specifically addressing conditions like corneal damage. This innovative surgical technique relies on stem cell-based treatments to regenerate the protective layer of the cornea, promoting healing in cases that were previously deemed irreparable. By leveraging the regenerative qualities of limbal epithelial cells, this novel procedure showcases the potential of advanced cellular therapies in restoring vision and improving the quality of life for patients. As the clinical data continues to reveal positive outcomes, CALEC emerges as a pivotal development in eye care, bridging the gap between existing treatments and revolutionary methods aimed at combating visual impairments.
Introduction to CALEC Surgery and Its Significance
The emergence of the Cultivated Autologous Limbal Epithelial Cell (CALEC) surgery marks a significant advancement in ophthalmology, particularly for patients suffering from severe corneal damage. CALEC surgery involves a sophisticated technique where stem cells from a healthy eye are harvested, cultured, and then transplanted into the affected eye, effectively restoring its surface. This procedure embodies hope for those with previously untreatable corneal injuries, providing a new avenue for eye damage treatment that comprehensively addresses the issue of limbal stem cell deficiency, a condition that severely compromises vision.
At Mass Eye and Ear, the pioneering work of Ula Jurkunas and her team highlights the collaborative nature of this surgery, integrating advanced stem cell therapy with clinical expertise. The initial trial showcased a remarkable success rate, with over 90% of treated patients experiencing substantial improvements in their corneal surface. Such outcomes not only enhance visual acuity but also significantly improve the quality of life for individuals who previously faced the prospect of irreversible vision loss. This innovative surgery is a testament to the evolving field of regenerative medicine and its potential applications in eye care.
The Process of CALEC Surgery and Stem Cell Therapy
The CALEC surgery process involves meticulous steps beginning with the extraction of limbal epithelial cells from a healthy donor eye via a biopsy. These cells are cultivated in a specialized environment to create a tissue graft, a process that typically spans two to three weeks. This innovative manufacturing technique is crucial, as it enables the generation of enough viable cells to effectively restore the damaged corneal surface of the recipient patient. By utilizing the body’s own stem cells, CALEC surgery represents a natural and biocompatible approach to one of the most challenging problems in ophthalmology.
During the clinical trial at Mass Eye and Ear, participants underwent careful monitoring to evaluate the effectiveness and safety of this groundbreaking intervention. By utilizing advanced protocols, the research team ensured that each graft met the stringent quality standards required for human transplant. The results of this trial demonstrate not only the high success rate of the procedure in restoring vision but also the minimal incidence of adverse events, establishing CALEC as a promising avenue for addressing severe corneal injuries.
The Role of Limbal Epithelial Cells in Corneal Health
Limbal epithelial cells play a pivotal role in maintaining the health of the corneal surface. These specialized stem cells reside in the limbus, the boundary zone between the cornea and the sclera, and are responsible for the continuous regeneration of the corneal epithelium. When trauma occurs—due to chemical burns, infections, or physical injuries—the loss of these cells leads to significant complications, including persistent pain and visual impairment. The depletion of limbal epithelial cells creates a challenge for traditional corneal transplant techniques, often leaving patients without viable treatment options.
Within the context of CALEC surgery, the ability to restore these essential cells offers hope for many individuals facing debilitating eye damage. By harnessing the regenerative capabilities of stem cells, CALEC surgery not only repairs the damaged corneal surface but also provides the potential for long-lasting improvement in vision. This innovative approach signifies a shift towards personalized and precise treatment options in ophthalmic care, emphasizing the importance of cellular health in the overall function of the eye.
Clinical Outcomes and Future Directions in CALEC Research
The clinical outcomes of the CALEC trial reflect its promise as a viable treatment for patients with severe corneal damage. Initial findings indicate that 50% of participants achieved complete restoration of their corneal surface within three months, with success rates climbing to 79% and 77% at the 12- and 18-month follow-ups, respectively. This significant data reinforces the potential for CALEC surgery to become a standard treatment option for those suffering from limbal stem cell deficiency, thereby broadening accessibility to effective eye damage treatment.
As the research progresses, future CALEC studies aim to incorporate larger patient cohorts and diversify the research across multiple medical centers. This broader approach will facilitate a more robust understanding of the procedure’s efficacy and safety profile, further supporting its pathway toward FDA approval. Researchers are also exploring the possibility of an allogeneic manufacturing process for CALEC, which would involve sourcing stem cells from cadaveric donors, potentially expanding the treatment options for patients with bilateral eye damage. The development of these advancements highlights the ongoing commitment within the medical community to enhance regenerative therapies for ocular health.
Safety Profile of CALEC Surgery
One of the significant advantages of the CALEC surgery trial is its impressive safety profile. With no serious adverse events recorded among participants, this procedure underscores the thoroughness of the research and the commitment to patient well-being. Minor issues, such as a single case of a bacterial infection attributed to chronic contact lens use, emphasize the importance of post-operative care. This careful monitoring not only reassures patients about the risks involved but also showcases the rigorous protocols established by the research team at Mass Eye and Ear.
In light of these outcomes, the safety and effectiveness of stem cell therapies like CALEC must be continuously evaluated. As additional trials are planned, ongoing assessments will focus on long-term effects and potential complications, ensuring that any new treatments will not just be cutting-edge but also safe for widespread clinical application. The continued emphasis on safety will be vital in fostering trust within the medical community and among patients as these innovative solutions become more prevalent in ophthalmology.
Unpacking the Scientific Collaboration Behind CALEC
The success of CALEC surgery can be attributed to extensive scientific collaboration among leading institutions. Mass Eye and Ear, along with partners such as Dana-Farber Cancer Institute, has worked tirelessly to advance stem cell therapy for ocular applications. This teamwork is critical as it brings together diverse expertise, from cell manufacturing to clinical application, ensuring that the final treatment is both efficacious and safe for patients. The innovative spirit of this collaboration demonstrates how a multidisciplinary approach can lead to groundbreaking solutions in eye care.
In particular, collaboration with experts in regenerative medicine, as seen in the contributions from Jerome Ritz and his team, highlights the importance of synergy in research. By combining resources and knowledge, the team has been able to meet the rigorous quality assurance standards required for transplant-ready stem cells. Such strategic alliances not only expedite the process of bringing new therapies to clinical application but also foster an environment of shared knowledge and expertise that can lead to further innovations in the treatment of eye diseases.
The Importance of Regulatory Approval for CALEC Treatments
As CALEC surgery represents a novel treatment modality in ophthalmology, navigating the path toward regulatory approval is essential. The trial conducted at Mass Eye and Ear, deemed the first of its kind in the United States to apply stem cell therapy for ocular treatment, exemplifies the substantial consideration given to the safety and efficacy of new treatments. Gaining approval from the U.S. Food and Drug Administration (FDA) involves rigorous evaluation of clinical data, and this process is vital to ensure that future applications of CALEC can be broadly accessible to patients in need.
The ongoing commitment to meet regulatory standards illustrates the dedication of researchers like Ula Jurkunas and her colleagues to provide a credible foundation for this revolutionary therapy. As data from the trials continues to demonstrate the efficacy and minimal risks associated with CALEC surgery, hopes for successful approval heighten. This milestone not only represents a victory for researchers and clinicians but also an enormous leap forward in the treatment of previously untreatable corneal injuries.
Patient Perspectives on CALEC Surgery Outcomes
Understanding the impact of CALEC surgery from the patient perspective is crucial in evaluating its overall success. Many participants in the clinical trial have reported significant improvements in their vision and quality of life following the procedure. Patients who once lived with the burden of severe corneal injuries found renewed hope and functionality thanks to the restoration achieved through stem cell therapy. This first-hand experience highlights the transformative potential of CALEC surgery, reaffirming its role in redefining outcomes for individuals dealing with eye damage.
Moreover, as conversations around patient outcomes continue, it emphasizes the importance of shared decision-making in clinical settings. Patients engaged in discussions about their treatment options expressed a desire for innovative therapies like CALEC, showcasing their interest in participating in cutting-edge clinical trials. This feedback not only serves to validate the therapeutic advancements being made at Mass Eye and Ear but also highlights the essential role patient experiences play in shaping future research and development in the field of ophthalmology.
Future Prospects for Stem Cell Therapy in Ophthalmology
The successful execution of CALEC surgery at Mass Eye and Ear is paving the way for future advancements in stem cell therapy, particularly within the realm of novel ocular treatments. As researchers refine the methodologies associated with CALEC and explore allogeneic options, the potential applications of stem cell therapy in ophthalmology will expand significantly. This innovation could revolutionize the treatment landscape, offering hope to a broader range of patients facing diverse ocular complications.
Additionally, the insights gained from ongoing CALEC studies will likely facilitate advancements in related fields, encouraging other researchers to explore stem cell applications for various eye diseases. As more data becomes available, it is anticipated that stem cell therapy might form the cornerstone of future ocular regenerative medicine strategies, ultimately transforming the prognosis for countless individuals affected by corneal damage and other related disorders.
Frequently Asked Questions
What is CALEC surgery and how does it relate to eye damage treatment?
CALEC surgery, or cultivated autologous limbal epithelial cells surgery, is an innovative treatment developed at Mass Eye and Ear that utilizes stem cell therapy to restore the corneal surface in patients with severe eye damage. By harvesting limbal epithelial cells from a healthy eye, expanding them into a cellular graft, and transplanting them into the damaged eye, CALEC surgery aims to repair corneal defects that conventional treatments cannot address.
How does stem cell therapy work in CALEC surgery?
In CALEC surgery, stem cell therapy involves taking limbal epithelial cells from a healthy eye and cultivating them into a tissue graft. This graft is then transplanted into the affected eye to restore the cornea’s surface. The process not only repairs damage but also promotes healing where traditional methods have failed.
Who is eligible for CALEC surgery at Mass Eye and Ear?
Eligibility for CALEC surgery typically includes patients who have a damaged cornea due to injuries like chemical burns or infections, and who possess one healthy eye from which limbal stem cells can be harvested. The specific criteria are determined by the medical team at Mass Eye and Ear.
What are the success rates of CALEC surgery in clinical trials?
Clinical trials for CALEC surgery have demonstrated impressive outcomes, showing over 90% effectiveness in restoring the corneal surface in patients. Follow-up results indicated that about 79% to 93% of participants experienced complete or partial success, significantly improving their vision.
What are the potential risks associated with CALEC surgery?
While CALEC surgery has shown a high safety profile, potential risks include minor adverse events such as infections, particularly related to contact lens use. However, serious complications have not been reported in the studied patients. Continuous monitoring is essential to manage any such risks during and after the procedure.
Why is CALEC surgery considered a significant advancement in corneal damage treatment?
CALEC surgery is revolutionary because it introduces a stem cell-based approach that can restore the cornea in cases previously deemed untreatable. It represents a paradigm shift in ocular treatments, allowing for the repair of severe corneal injuries through biological regeneration rather than reliance on transplant or artificial materials.
How long does the CALEC procedure take from start to finish?
The entire CALEC surgery process, including the biopsy to collect limbal epithelial cells, expansion of the cells into a graft, and the actual transplant surgery, typically spans two to three weeks for preparation before the surgical procedure can be performed.
Is CALEC surgery currently available for patients outside of clinical trials?
Currently, CALEC surgery is still in the experimental phase and is not widely available, even at Mass Eye and Ear. Additional trials and research are necessary before it can be offered as a standard treatment for corneal damage in a clinical setting.
What are the future prospects for CALEC surgery and similar treatments?
Future studies aim to broaden the application of CALEC surgery, potentially including allogeneic grafts from cadaveric donors. This advancement could help treat patients with corneal damage in both eyes and enhance the accessibility of such promising stem cell therapies.
Who conducted the CALEC surgery clinical trials, and what organizations supported the research?
The clinical trials for CALEC surgery were led by Ula Jurkunas and Reza Dana at Mass Eye and Ear, with support from the National Eye Institute, part of the National Institutes of Health. These collaborative efforts included research teams from Dana-Farber and Boston Children’s Hospital.
| Key Points |
|---|
| Ula Jurkunas conducted the first CALEC surgery, introducing a new hope for treating corneal damage once deemed untreatable. |
| The CALEC procedure involves stem cell therapy where stem cells are harvested from a healthy eye and used to repair a damaged cornea. |
| In a clinical trial, CALEC restored the corneal surface in 14 patients over an 18-month period, with over 90% effectiveness. |
| The trial involved a biopsy of an unaffected eye, and CALEC can currently only be used if only one eye is damaged. |
| Future studies aim to use cadaveric donor eyes to expand treatment to patients with damage in both eyes. |
| The procedure has shown a high safety profile, with minimal adverse events reported. |
| Continued research and larger trials are necessary before CALEC surgery can receive FDA approval. |
| The study was the first human trial of a stem cell therapy funded by the National Eye Institute. |
Summary
CALEC surgery marks a groundbreaking advancement in corneal treatment, presenting an innovative solution for patients facing previously untreatable injuries. The study conducted by Ula Jurkunas and her team at Mass Eye and Ear demonstrated the promising efficacy and safety of the cultivated autologous limbal epithelial cell therapy. By harnessing the body’s own stem cells to repair corneal damage, CALEC surgery not only restores vision but also significantly improves patients’ quality of life. As research continues, the hope is to expand access to this revolutionary treatment, paving the way for those with bilateral corneal injuries in the future.